|
Hence, this salt water certainly has a density of 1.1 g/mL. Step 2: Putting these values into the formula 
Mass of salt water = mass salt + mass of waterĪlso, the volume of salt water is 200 mL. Now, one can find out the mass of the saltwater Mass of water = 1 g/mL multiplied by 200 mL Find out the density of the salt water? (note = density of water is 1 g/mL)Īnswer: Step 1: Derive the mass and volume of the salt water.ĭensity of water = mass of water/volume of water Calculating density using grams and cubic centimetres would give a density unit of grams per cubic centimetre (g/cm³). A particular solution of salt water contains 20 grams of salt and 200 grams of water. Hence, this sugar cube has a density of 1.5 grams/cm 3. Step 2: Putting the variables into the formula The volume of a cube = (length of side) 3 Step 1: Derive the mass and volume of this sugar cube 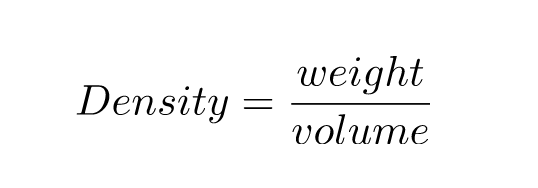
How do you calculate surface density Area or surface density is the amount of a quantity (often mass) per unit of area. Density is commonly expressed in units of grams per cubic centimetre. Find out the density of a cube of sugar which weighs 12 grams and measures 2 cm on a side?Īnswer: This question can be completed in two steps The formula for density is d M/V, where d is density, M is mass, and V is volume. Here, ρ = density, m = mass, and v = volume. Some individuals also make use of the letter D for expressing this measurement. Furthermore, this symbol happens to be the lower case Greek letter rho. Density Formula: m/V, where is the density, m is the mass of the object and V is the volume of the object. Most noteworthy, the symbol ρ is mostly made use of for expressing this measurement. The definition of this measure can be expressed as mass, m, in a particular volume, v. Density Formulaĭensities refer to the measure of relative compactness. Heating the bottom of a liquid in most cases results in a decrease in this measurement of such heated liquid. This ultimately causes an increase in this particular measurement of the object.Īlso, increasing a substance’s temperature results in a decrease in this measurement. When the pressure on an object increases, then consequently the volume decreases. This variety is comparatively small for solids and liquids but significantly greater when it comes to gases. The densities of materials show variation with pressure and temperature. Relative densities refer to the ratio of a substance’s density to a standard substance or material, usually water. 
The replacement of this measurement with a concept is known as relative density can sometimes take place. One can simplify this measurement’s comparisons across the different systems of units. Moreover, this particular measurement of a material can be relevant to purity, buoyancy, and packaging. Densities vary with different materials or substances. This measurement of a pure substance has the same value as its mass concentration. 3 Solved Example on Density Formula What is Density?ĭensity refers to the measurement of the amount of mass of a substance per unit of volume.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
